Hepatitis C virus (HCV) is hyperendemic among people who inject drugs (PWID), with prevalence rates typically ranging between 40% and 90%. HCV results in cirrhosis, liver failure, and hepatocellular carcinoma for a substantial proportion of those infected. Yet treatment for HCV infection is costly, and PWID are less likely to receive medical monitoring and treatment of the infection than other patient groups. Beginning in 2007, there were more deaths due to HCV than HIV in the U.S. Thus, the prevention of primary HCV infection among PWID is a public health issue of major importance, and behavioral and structural interventions are needed to prevent HCV transmission among PWID.
This Scientific Brief summarizes a recent systematic review and meta-analysis conducted by CDUHR Investigator Dr. Holly Hagan and colleagues of the association between HCV seroconversion and interventions that are intended to reduce injection-related acquisition of HCV among PWID, followed by the study’s implications for policy.
- Data for these analyses were collected as part of the HCV Synthesis Project, a systematic review and meta-analysis of HCV epidemiology and prevention in drug users (see Stern et al., 2008 for details on the methods).
- The scope of the HCV Synthesis Project includes published and unpublished reports describing the epidemiology of HCV infection in PWID and in non–injection drug users who sniff or smoke heroin, cocaine, or amphetamine.
- Research reports were included in the analysis if they reported HCV seroconversion rates in PWID in relation to interventions that could, in theory, prevent HCV infection by reducing unsafe injections.
- These interventions included drug treatment programs, syring access programs, supervised injection facilities, syringe disinfection with bleach, behavioral interventions, or combinations of any of these services, all measured at the individual level. For each type of intervention, we pooled the data and calculated overall estimates of its effect on HCV seroconversion.
- A total of 26 reports (4 randomized controlled trials and 22 observational studies) published between January 1989 and April 2010 met inclusion criteria.
Interventions that combined substance-use treatment and support for safe injection reduced HCV seroconversion by 75%. Single-intervention strategies such as substance-use treatment or syringe access did not prevent HCV infection.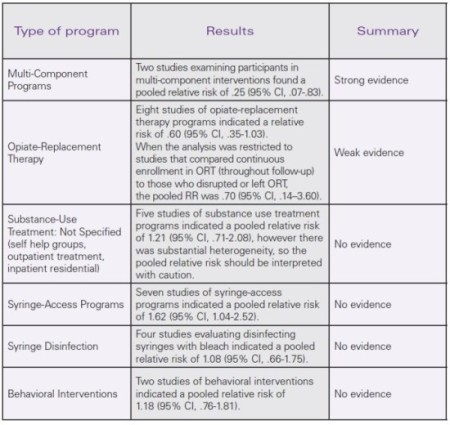
- The current available research indicates that HCV can be prevented among PWID.
- Combination prevention strategies are associated with a substantial and statistically significant reduction in HCV incidence in PWID.
- This finding is consistent with the understanding that an array of factors facilitate HCV transmission among PWID, including the high prevalence of HCV-infectious injectors and the efficiency with which HCV may be transmitted via a number of different drug injection-related practices.
- Thus, multi-component interventions that support a range of strategies (such as reduction or elimination of drug injection, adoption of safe injection practices through the provision of sterile syringes and drug-preparation equipment, or behavior-change counseling) would be expected to achieve greater success than those offering fewer options for lowering risk.
- Both of the multi-component interventions examined here included opioid replacement therapy (ORT). However, the meta-analysis of ORT alone showed a less substantial and inconsistent impact on HCV seroconversion risk. Therefore, one cannot conclude that the large-magnitude effect of multi-component programming on HCV infection rates is wholly attributable to ORT.
- Multi-component interventions that include methods to reduce drug-use frequency and to support safe injection should be developed and tested in a variety of settings and subsets of PWID (e.g., among young and racial or ethnic minority PWID, and in low- and middle-income countries).
- As with all meta-analyses, the study was restricted to the data that could be obtained from written reports.
- Weak effects found in some studies that were included in the meta-analysis may have been due to insufficient participation in the intervention, for example, participants who enrolled in ORT for a short period of time.
- Structural interventions, such as syringe-access programs, are intended to reduce infection in a population of PWID. Individual-level comparisons of syringe access program users to non-users are typically biased because they attract and retain higher-risk PWIDs (Hagan & Thiede, 2000).
Hagan H, Pouget ER & Des Jarlais DC (2011). A systematic review and meta-analysis of interventions to prevent hepatitis C virus infection in people who inject drugs. Journal of Infectious Diseases, 204(1), 74-83.
Hagan H (2011). Agent, host, and environment: hepatitis C virus in people who inject drugs. Journal of Infectious Diseases, 204(12), 1819-1821.
Hagan H & Thiede H (2000). Changes in injection risk behavior associated with participation in the Seattle needle-exchange program. Journal of Urban Health, 77(3), 369-382.
Stern RK, Hagan H, Lelutiu-Weinberger C, Des Jarlais DC, Scheinmann R, Strauss S, Pouget ER & Flom P (2008). The HCV Synthesis Project: Scope, methodology, and preliminary results. BMC Medical Research Methodology, 8, 62.
Contact: Holly Hagan, PhD at hh50@nyu.edu
Supported by a grant from the National Institute on Drug Abuse (R01 DA018609) and the NIDA-funded Center for Drug Use and HIV Research (CDUHR; P30 DA121041).